Comparative Effectiveness Research: Patient-Centered Outcomes Research Institute and HHS Continue Activities and Plan New Efforts
Fast Facts
The Patient-Centered Outcomes Research Institute is a federally funded, nonprofit corporation that funds and evaluates research aimed at improving health care decisions.
Among other things, the Institute funds research to compare available treatment options for various diseases and conditions. The Institute—and Health and Human Services—are also required to share the findings of the research.
The Institute met its goal of getting its research cited in more news and online sources. HHS took steps to assess its effectiveness in sharing findings and getting them implemented. Both plan to comprehensively evaluate their efforts in the future.

Highlights
What GAO Found
GAO found that the Patient-Centered Outcomes Research Institute (PCORI)—a federally funded, nonprofit corporation—and the Department of Health and Human Services (HHS) have continued to perform comparative clinical effectiveness research (CER) activities required by law since our prior report issued in 2015. CER evaluates and compares health outcomes, risks, and benefits of medical treatments, services, or items. The requirements direct PCORI and HHS to, among other things, fund CER and disseminate and facilitate the implementation of CER findings.
GAO's analysis of PCORI and HHS documents show that they allocated a total of about $3.6 billion for CER activities and program support during fiscal years 2010 through 2019 from the Patient Centered Outcomes Research Trust Fund (Trust Fund). Specifically, PCORI allocated about $2 billion for research awards and another $542 million for other awards, to be paid over multiple years. HHS allocated about $598 million for activities such as the dissemination and implementation of CER findings. PCORI and HHS also allocated about $470 million for program support.
PCORI and HHS Allocations for Comparative Clinical Effectiveness Research (CER) Activities, Fiscal Years 2010 through 2019
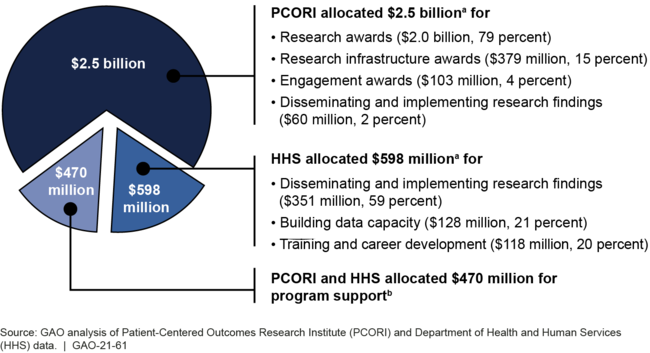
aTotals may not add up due to rounding.
bPCORI and HHS allocated $457 million and $13 million for program support, respectively.
PCORI assessed the effectiveness of its activities using performance measures and targets. Since fiscal year 2017, when early CER projects were completed, PCORI officials reported that the institute met its performance targets, such as an increased number of research citations of its CER findings in news and online sources. HHS described accomplishments or assessed the effectiveness of its dissemination and implementation activities. PCORI and HHS officials told GAO they are planning comprehensive evaluations of their CER dissemination and implementation activities as part of their strategic plans for the next 10 years.
Why GAO Did This Study
The 2010 Patient Protection and Affordable Care Act (PPACA) authorized establishment of PCORI to conduct CER and improve its quality and relevance. PPACA also established new requirements for HHS to, among other things, disseminate findings from federally funded CER and coordinate federal programs to build data capacity for this research. To fund CER activities, PPACA established the Trust Fund, which provided a total of about $3.6 billion to PCORI and HHS for CER activities during fiscal years 2010 through 2019. The Further Consolidated Appropriations Act, 2020, added new CER requirements and extended funding at similar levels through fiscal year 2029.
PPACA and the Appropriations Act 2020 included provisions that GAO review PCORI and HHS's CER activities. This report describes (1) the CER activities PCORI and HHS carried out to meet legislative requirements, (2) how PCORI and HHS allocated funding to those CER activities, and (3) PCORI and HHS efforts to evaluate the effectiveness of their CER dissemination and implementation activities, such as changes in medical practice.
GAO reviewed legislative requirements and PCORI and HHS documentation and data for fiscal years 2010-2019. GAO also interviewed PCORI and HHS officials and obtained information from nine selected stakeholder groups that were familiar with PCORI's or HHS's CER activities. These groups included payer, provider, and patient organizations. GAO incorporated technical comments from PCORI and HHS as appropriate.
For more information, contact John Dicken at (202) 512-7114 or dickenj@gao.gov.
