High-Containment Laboratories: Actions Needed to Mitigate Risk of Potential Exposure and Release of Dangerous Pathogens
Fast Facts
In 2015, the Department of Defense discovered that one of its labs had inadvertently sent live anthrax to almost 200 other labs worldwide over 12 years. The lab's "inactivation" (removal of hazardous effects) of the pathogen was incomplete.
Both the science and the federal guidance around pathogen inactivation are limited and inconsistently implemented. No one knows how many incomplete inactivation incidents have occurred because labs don't have to identify them in incident reports, and are only required to report incidents involving certain pathogens. The report related to this testimony recommends ways to address incomplete inactivation risks.
Sites That Received Viable Anthrax Samples Thought to be Inactivated from the Department of Defense’s Dugway Proving Ground, during 2004 through 2015
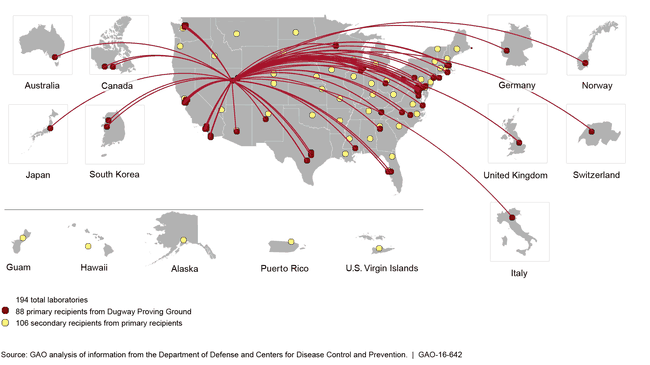
Map showing locations of 194 laboratories (88 primary recipients and 106 secondary recipients).
Highlights
What GAO Found
The total number of incidents involving incomplete inactivation--a process to destroy the hazardous effects of pathogens while retaining characteristics for future use--that occurred from 2003 through 2015 is unknown for several reasons. One key reason is that the Select Agent Program--operated by the Departments of Health and Human Services (HHS) and Agriculture (USDA) to oversee certain dangerous pathogens, known as select agents--does not require laboratories to identify such incidents on reporting forms. According to the program, 10 incidents occurred from 2003 through 2015. However, GAO identified an additional 11 incidents that the program did not initially identify. Because the program cannot easily identify incidents involving incomplete inactivation, it does not know the frequency or reason they occur, making it difficult to develop guidance to help mitigate future incidents. The 21 identified incidents involved a variety of pathogens and laboratories, as shown below.
Figure: Twenty-one Identified Incidents Involving Incomplete Inactivation that Occurred from 2003 through 2015 by Pathogen and Laboratory Type

Several challenges affect the implementation of inactivation in high-containment laboratories, including gaps in scientific knowledge and limited guidance. For example, there is limited federal guidance for researchers on the development and validation of inactivation protocols. Validation helps ensure protocols are scientifically sound and produce consistent results. Due to limited guidance, laboratories varied in their interpretation of validated methods of inactivation, resulting in researchers applying differing levels of rigor. Without more comprehensive guidance, as called for by experts, protocols will vary in their scientific soundness, increasing the risk of incomplete inactivation.
The Select Agent Program did not consistently refer incidents involving incomplete inactivation for further investigation and enforcement for violations of select agent regulations. For example, the program referred incidents involving incomplete inactivation at various laboratories, but did not refer two incidents in 2014 that occurred at HHS. A memorandum of understanding between HHS and USDA states that the program should handle incidents consistently. GAO found, however, that the program does not have a consistent, written set of criteria for handling incidents. Without such criteria, the program risks inconsistent enforcement of select agent regulations. This further highlights GAO's previous finding that existing federal oversight of high-containment laboratories is fragmented and self-policing.
Why GAO Did This Study
This testimony summarizes the information contained in GAO's September 2016 report, entitled High-Containment Laboratories: Improved Oversight of Dangerous Pathogens Needed to Mitigate Risk (GAO-16-642).
For more information, contact Timothy M. Persons, Ph.D. at (202) 512-6412 or personst@gao.gov or John Neumann at (202) 512-3841 or neumannj@gao.gov.
