E-cigarettes and FDA’s New Rule
E-cigarettes have rapidly become more popular since they first appeared on the U.S. market about 10 years ago. Just last week, the Food and Drug Administration issued a rule to regulate e-cigarettes as tobacco products.
This follows recent e-cigarette legislation requiring liquid nicotine packaging to be child-resistant and meet other poison prevention standards. As the legal landscape surrounding these relatively new products has changed, the WatchBlog has been keeping an eye on e-cigarettes.
Today’s post explores e-cigarette regulation and taxes—and why we love good data.
New product, new rules
In the new final rule released on May 5, 2016, FDA extended its tobacco product regulation authority to cover e-cigarettes and other products that meet the statutory definition of “tobacco product”—such as cigars, pipe tobacco, hookah, and nicotine gels.
According to FDA, the rule prohibits the sale of “covered tobacco products,” including e-cigarettes, to individuals under the age of 18 and requires health warning labels on packages and in advertisements. Moreover, FDA stated that going forward it will be able to:
- review new tobacco products not yet on the market,
- help prevent misleading claims by tobacco product manufacturers,
- evaluate the ingredients of tobacco products and how they are made, and
- communicate the potential risks of tobacco products.
A taxing question
According to FDA’s new rule, neither the action FDA has taken under the rule nor any other FDA regulations directly affect the taxation of any tobacco product. At present, the Internal Revenue Code of the United States, which sets federal tax rates, does not specifically define or list a tax rate for e-cigarettes.
And while e-cigarette use is increasing, federal tax revenue from traditional cigarettes has been declining.
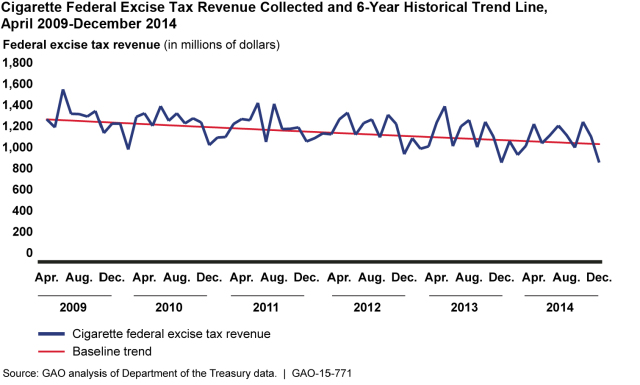 (Excerpted from GAO-15-771)
(Excerpted from GAO-15-771)
This graph raises the question of whether e-cigarette use contributed to the decline, but from the data available in 2015 we couldn’t measure any effect of e-cigarettes on the traditional cigarette market. Reasons for that include:
- the relatively small size of the e-cigarette market—estimated at $2.5 billion in 2014—compared to the traditional cigarette market, which was $80 billion in the same year;
- lack of good data about whether smokers are substituting e-cigarettes for cigarettes; and
- limited data on e-cigarette quantities and prices.
Better data = better understanding
To help defray the costs of expanding oversight to additional tobacco products, FDA may propose another new rule to impose user fees on e-cigarette manufactures and importers. If it does, FDA may start collecting data on quantities of e-cigarettes sold on the U.S. market to calculate the fees.
Starting January 1, 2016, the federal government started collecting data on e-cigarette imports. These data and the potential new data from FDA could, in a few years, mean more and better federal data on U.S. e-cigarette quantities. These data could help both regulators and researchers analyze the U.S. e-cigarette market, e-cigarette use, and the effects of e-cigarette use on the traditional cigarette market.
- Comments on GAO’s WatchBlog? Contact blog@gao.gov.
GAO Contacts
Related Products

GAO's mission is to provide Congress with fact-based, nonpartisan information that can help improve federal government performance and ensure accountability for the benefit of the American people. GAO launched its WatchBlog in January, 2014, as part of its continuing effort to reach its audiences—Congress and the American people—where they are currently looking for information.
The blog format allows GAO to provide a little more context about its work than it can offer on its other social media platforms. Posts will tie GAO work to current events and the news; show how GAO’s work is affecting agencies or legislation; highlight reports, testimonies, and issue areas where GAO does work; and provide information about GAO itself, among other things.
Please send any feedback on GAO's WatchBlog to blog@gao.gov.



