Drug Safety: Preliminary Findings Indicate Persistent Challenges with FDA Foreign Inspections
Fast Facts
More than 60% of drug manufacturers for the U.S. market are located overseas. The Food and Drug Administration inspects foreign and domestic drug manufacturers to ensure drug safety and effectiveness.
Among other things, we testified that:
The number of inspections of foreign drug manufacturers has declined since FY 2016, which FDA attributes in part to fewer inspectors and difficulty filling jobs abroad
Most foreign inspections are preannounced, giving manufacturers time to prepare for inspections
We have long-standing concerns about FDA’s ability to oversee the global supply chain, a High Risk Series issue for 10 years.

Prescription drugs
Highlights
What GAO Found
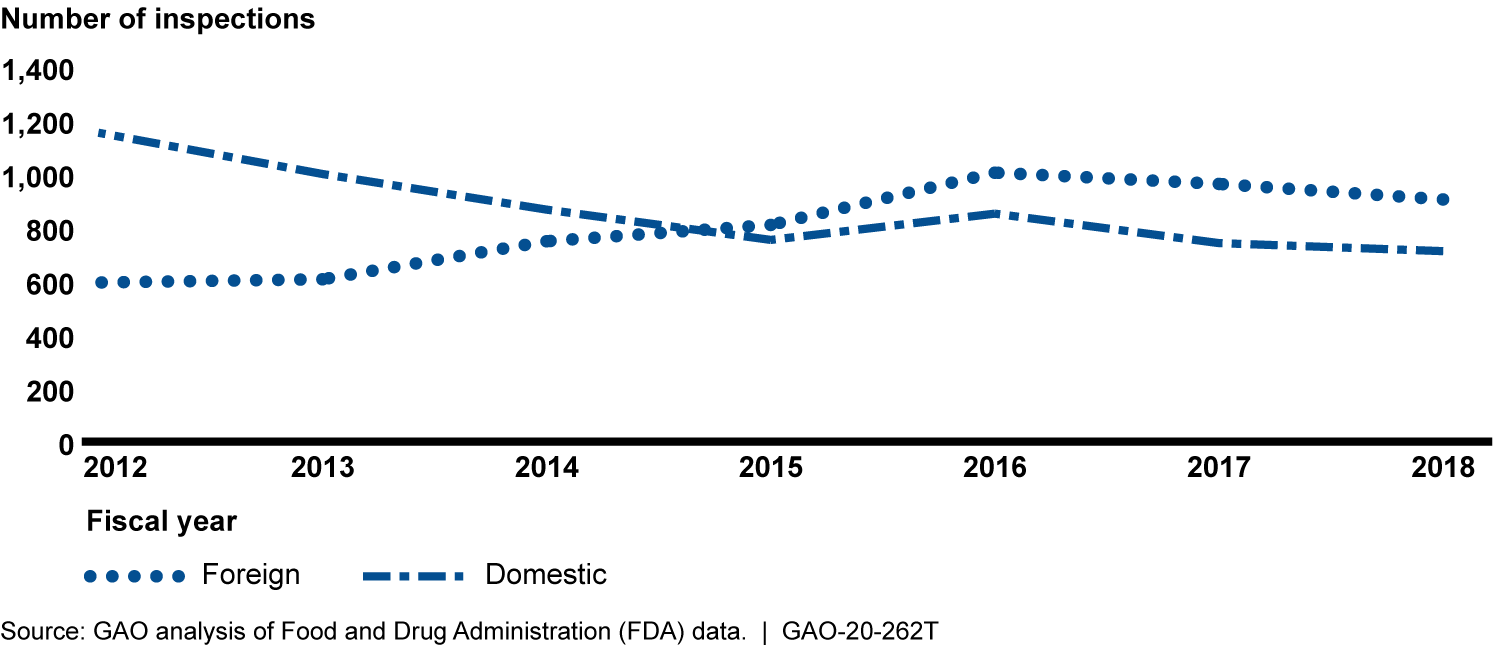
GAO's preliminary analysis of Food and Drug Administration (FDA) data shows that from fiscal year 2012 through 2016, the number of foreign drug manufacturing establishment inspections increased. From fiscal year 2016 through 2018, both foreign and domestic inspections decreased—by about 10 percent and 13 percent, respectively. Howevever, the total number of foreign inspections surpassed the number of domestic inspections in 2015, and a growing percentage of FDA's foreign inspections (43 percent in 2018) were conducted in China and India, where most establishments that ship drugs to the United States were located. FDA officials attributed the decline, in part, to vacancies among investigators available to conduct inspections. GAO previously noted the vital role that inspections play in FDA's oversight of foreign establishments.
FDA Inspections of Foreign and Domestic Drug Establishments, Fiscal Year 2012 through 2018
FDA has vacancies among each of the groups of investigators who conduct foreign inspections. FDA had 190 investigators in the United States who conduct the majority of foreign inspections, but an additional 58 positions were vacant. FDA was in the process of filling 26 of these vacancies, with 32 remaining. However, according to FDA officials, it could be 2 to 3 years before new staff are experienced enough to conduct foreign inspections. FDA also faces persistent vacancies among investigators in its foreign offices.
FDA investigators identified persistent challenges conducting foreign inspections, raising questions about the equivalence of foreign to domestic inspections. For example, while domestic inspections are almost always unannounced, FDA's practice of preannouncing foreign inspections up to 12 weeks in advance may give manufacturers the opportunity to fix problems. Investigators from FDA's China and India offices do conduct some unannounced inspections, but they are involved in a small percentage of inspections in these countries (27 percent and 10 percent, respectively). Further, FDA continues to rely on translators provided by the foreign establishments being inspected, which investigators said can raise questions about the accuracy of information FDA investigators collect.
Why GAO Did This Study
More than 60 percent of establishments manufacturing drugs for the U.S. market were located overseas in fiscal year 2018. FDA has estimated that about 40 percent of finished drugs and 80 percent of active drug ingredients are manufactured overseas. FDA is responsible for overseeing the safety and effectiveness of all drugs marketed in the United States, regardless of where they are produced and conducts inspections of both foreign and domestic drug manufacturing establishments. GAO has had long-standing concerns about FDA's ability to oversee the increasingly global supply chain, an issue highlighted in GAO's High Risk Series for the last 10 years. GAO recommended in 2008 (GAO-08-970) that FDA increase the number of inspections of foreign drug establishments. GAO found in 2010 (GAO-10-961) and 2016 (GAO-17-143) that FDA was conducting more of these foreign drug inspections, but GAO also reported that FDA may have never inspected many establishments manufacturing drugs for the U.S. market.
This statement is based on ongoing work and provides preliminary GAO observations on 1) the number of foreign inspections FDA has conducted, 2) inspection staffing levels, and 3) challenges unique to foreign inspections. For this work, GAO examined FDA data, visited FDA foreign offices in China and India, and interviewed drug investigators based in these offices and in the United States.
Recommendations
GAO will continue to assess these issues as part of ongoing work, and make recommendations as appropriate.
