DOD Health Care: Defense Health Agency Should Improve Tracking of Serious Adverse Medical Events and Monitoring of Required Follow-up
Fast Facts
Medical errors, system vulnerabilities, or process failures can permanently or temporarily harm—or kill—patients. In military health facilities, it's critical to track and analyze these "sentinel events" to help prevent them in the future.
The military services and the Defense Health Agency both track such events. Agency officials told us they spend 80 hours a month reconciling their records with the military services, but in some instances, the records don't match. The agency may be missing opportunities to make military health care safer. We recommended improving the tracking of sentinel events and follow-up requirements.
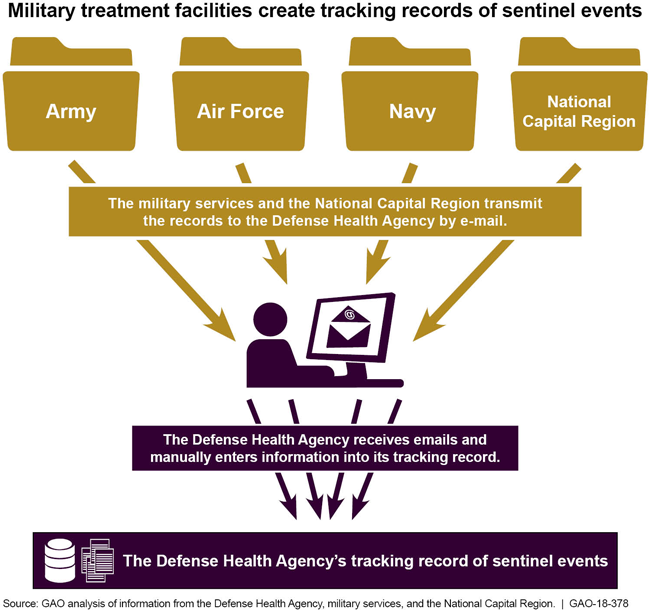
Army, Air Force, Navy & NCR send data to Defense Health Agency by email. DHA then manually enters into its tracking record.
Highlights
What GAO Found
GAO found that the process for tracking the most serious adverse medical events, called sentinel events, and their root cause analysis (RCA) reports are fragmented, impeding the Defense Health Agency's (DHA) ability to ensure that it has received complete information. Unlike other adverse medical events, sentinel events—which may result in severe harm or death—have additional reporting requirements that must be met within specified time frames. For example, military treatment facility (MTF) officials must develop RCA reports, which identify causal factors and corrective actions for sentinel events. However, because the database that DHA uses to collect information on adverse medical events does not currently have the capability to track this information, the military services (Army, Navy, and Air Force) and DHA each maintain their own tracking records for sentinel events and RCA reports. Due to these fragmented tracking efforts, DHA reconciles its information on sentinel events and RCA reports through monthly emails to the military services—a time-consuming, inefficient process. DHA officials emphasized that this process relies on the military services' cooperation because DHA does not currently have the authority to compel their responses. Moreover, despite DHA's reconciliation efforts, GAO identified discrepancies and missing information in DHA's tracking record. As a result, DHA lacks critical information about why a sentinel event may have occurred and what actions, if any, MTFs should take to prevent similar incidents in the future. Recently, DHA replaced its previous system of emails with a new tracker tool that can be accessed on the military health system website. However, the new tracker does not allow the military services to make edits, and as a result, any corrections or additional information must be submitted to DHA via email, which may perpetuate previous inefficiencies.
GAO found that DHA cannot ensure that it is receiving all reports on the implementation of corrective actions identified in RCA reports as required by a March 2015 memo. DHA officials stated that MTFs could meet this requirement by submitting copies of their measures of success (MOS) reports, which may be required by the Joint Commission, a hospital accrediting organization. As of September 2017, DHA had received 27 MOS reports for the 319 sentinel events that were reported in 2016. However, DHA does not know how many reports it is missing because MOS reports are not required for every sentinel event, and DHA did not began reconciling its information for these reports until January 2018, when it implemented its new tracker tool. Furthermore, GAO found that the new tracker tool documents the aggregate number of MOS reports received and does not indicate whether individual sentinel events have an MOS report, impeding DHA's ability to identify which reports are missing. This issue is compounded by the fact that the military services either track MOS reports in different ways or not at all, and military service officials said that DHA's requirement for MOS report submission is not clear. DHA officials stated that they expect to clarify this requirement in their update to the patient safety policy. Because it is unable to ensure it has received all reports on the implementation of corrective actions, DHA could be missing important information that could be used to help inform broader, system-wide patient safety improvement efforts.
Why GAO Did This Study
Adverse medical events are unintended incidents that may harm a patient. Serious adverse medical events, called sentinel events, have specific follow-up requirements. The National Defense Authorization Act for Fiscal Year 2017 (NDAA 2017) requires DHA to assume the military services' administrative responsibilities, such as adverse medical event reporting, for all MTFs beginning October 1, 2018.
The NDAA 2017 included a provision for GAO to examine the reporting and resolving of adverse medical events in the military health system. Among other objectives, this report reviews (1) the extent to which sentinel events and RCA reports are tracked and DHA ensures it has received complete information, and (2) the extent to which DHA ensures it has received MOS reports. GAO examined relevant policies; analyzed the most current available data on sentinel events from 2013 through 2016; and interviewed officials with DHA, the military services, and four MTFs selected for variety in military service, size, and geographic location.
Recommendations
GAO recommends that the Assistant Secretary of Defense (Health Affairs) ensure DHA (1) improve tracking of sentinel events and RCA reports, and (2) clarify its requirements for submitting reports on the implementation of corrective actions and consistently track and reconcile individual reports. DOD agreed with these recommendations.
Recommendations for Executive Action
| Agency Affected | Recommendation | Status |
|---|---|---|
| Office of the Assistant Secretary of Defense (Health Affairs) | The Assistant Secretary of Defense (Health Affairs) should ensure DHA improves as appropriate the systems and processes used by the military services, National Capital Region (NCR), and DHA to track sentinel events and RCA reports and require the military services and NCR to communicate with DHA the reasons RCA reports are not completed for reported sentinel events. (Recommendation 1) |
DOD concurred with this recommendation and has provided updates on its progress in implementing it. In December 2019, DOD reported that it had modified its policy on the systems and processes used to track sentinel events and their associated reports, and provided associated documentation. Changes include clarified reporting requirements, standardized templates for reporting sentinel events and associated follow up reports, and refinement of tracking tools. As such, we are closing this recommendation as implemented.
|
| Office of the Assistant Secretary of Defense (Health Affairs) | The Assistant Secretary of Defense (Health Affairs) should ensure DHA clarifies its requirement that reports on the implementation of corrective actions, such as MOS reports, should be completed and submitted to DHA, and to work with the military services and NCR to develop a standard system to help DHA consistently track and reconcile information about individual reports. (Recommendation 2) |
DOD concurred with this recommendation and has provided updates on its progress in implementing it. In June 2020, DOD provided documentation to show it has taken steps to address recommendation 2 through the implementation of policy changes, standardized templates, and refinement of tracking tools. Specifically, officials told us that since the publication of our report, they have fully implemented three tracking tools across the military services, which display all adverse medical events and their follow up reports across the military health system. They told us these tools replace the lengthy reconciliation process we described in our report. Additionally, officials told us adverse event records from all of the military services are visible in these tools, which motivates the military services to ensure their follow-up reports are submitted. Officials told us this has reduced the time the Defense Health Agency spends checking records and following-up on missing documentation by between 60 and 80%. As such, we are closing this recommendation as implemented.
|
