Emerging Animal Diseases: Actions Needed to Better Position USDA to Address Future Risks
Highlights
What GAO Found
The U.S. Department of Agriculture (USDA) did not take regulatory action during the initial response to the outbreaks of Swine Enteric Coronavirus Diseases (SECD) beginning in May 2013, when an outbreak was first detected, because the agency did not believe then that such action was necessary. Instead, USDA initially supported swine industry-led efforts. Without regulatory action, such as requiring reporting of infected herds, USDA had limited information about the location of the first infected herds. In addition, USDA officials acknowledged that USDA did not follow its guidance that calls for conducting epidemiological investigations at the onset of outbreaks. As a result, USDA did not conduct timely investigations of the premises with the first infected herds, and the source of disease will likely never be determined. Further, USDA does not have a process to help ensure the guidance is followed. Without such a process, USDA lacks reasonable assurance that the guidance will be followed in the future.
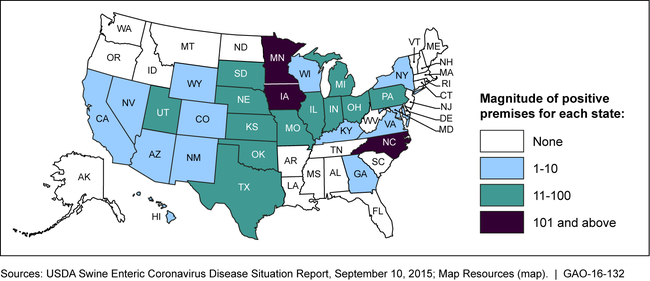
In June 2014, amid concerns about the spread of SECD, USDA issued a federal order requiring reporting of newly infected herds. As a result, USDA has more accurate information about the number and location of such herds, and SECD have been confirmed in 28 U.S. states, as shown below. USDA also provided funding to help manage the diseases.
Confirmed Swine Enteric Coronavirus Diseases from June 5, 2014 to September 5, 2015
To help improve its future response to SECD and other emerging animal diseases—those not known to exist in the United States or which have changed to become a threat—USDA has drafted new guidance. However, it has not defined key aspects of its response such as roles and responsibilities, which according to its strategic plan, are key components of successful collaboration to protect animal health. Without a clearly defined response to such emerging animal diseases, response efforts could be slowed.
Why GAO Did This Study
Pork is consumed more than any other meat worldwide, and there are numerous other products made with ingredients from pigs, including medical products, such as insulin to treat diabetes. The United States is the world's third-largest producer of pork products. USDA estimated that U.S. pork exports in 2014 were valued at over $6 billion. Two lethal, highly contagious diseases in pigs emerged in the United States in 2013 and 2014, causing the deaths of millions of pigs. The two emerging diseases are collectively known as SECD.
GAO was asked to review federal actions to address SECD outbreaks. This report examines (1) the initial response to the SECD outbreaks, (2) USDA's subsequent actions to manage SECD, and (3) steps USDA has taken to improve its future response to emerging animal diseases. GAO analyzed USDA efforts to collect data about the number and location of infected herds; reviewed federal regulations and USDA animal disease response guidance; and interviewed USDA, state, and industry stakeholders involved in the response and control efforts.
Recommendations
GAO recommends that USDA develop a process to help ensure its guidance for investigation of animal diseases is followed and clarify and document how it will respond to emerging diseases, including defining roles and responsibilities. USDA generally agreed with GAO's recommendations.
Recommendations for Executive Action
| Agency Affected | Recommendation | Status |
|---|---|---|
| Department of Agriculture |
Priority Rec.
To improve USDA's ability to respond to and protect against future emerging animal diseases, the Secretary of Agriculture should direct the Administrator of the Animal and Plant Health Inspection Service to clarify and document how the agency will respond to emerging diseases including defining key aspects of its response, such as roles and responsibilities, potential response strategies, and what may trigger different types of actions. |
USDA's Animal and Plant Health Inspection Service (APHIS) released its Emerging Animal Disease Preparedness and Response Plan in March 2017. In this plan, APHIS provides clarification on how it will respond to emerging animal diseases by outlining the process and responsibilities for identifying and characterizing the risks associated with threats to animal health, providing possible responses based on risk level, and describing communication and information sharing activities.
|
| Department of Agriculture |
Priority Rec.
To improve USDA's ability to respond to and protect against future emerging animal diseases, the Secretary of Agriculture should direct the Administrator of the Animal and Plant Health Inspection Service to develop a process to help ensure that its guidance for investigation of foreign or emerging animal diseases is followed, such as a process for documentation of the justification and approval of any deviation from the directions. |
USDA's Animal and Plant Health Inspection Service (APHIS) released its Emerging Animal Disease Preparedness and Response Plan in March 2017. This plan provides information on response options based on the assessed risk level of the animal disease, among other things. It also has a section that requires APHIS officials to document recommended response options that will then be reviewed and decisions made during regularly scheduled weekly or monthly meetings, or during ad hoc meetings, as needed, based on the urgency of the situation. This fulfills the intent of the recommendation.
|
