Medicare and Medicaid: Alignment of Managed Care Plans for Dual-Eligible Beneficiaries
Fast Facts
Eligible for both Medicare and Medicaid? In certain states you can get coverage from both programs through “aligned plans,” that is, private plans provided by the same or related companies. This arrangement is intended to better coordinate benefits and care delivery.
Some states support the automatic assignment of “dual-eligible” beneficiaries to aligned plans. Finding out more about these beneficiaries’ experiences could help the Centers for Medicare & Medicaid Services determine whether this practice poses any challenges to coordinating care.
We recommended collecting information on the experiences of beneficiaries assigned to aligned plans.

Stethoscope on a laptop keyboard
Highlights
What GAO Found
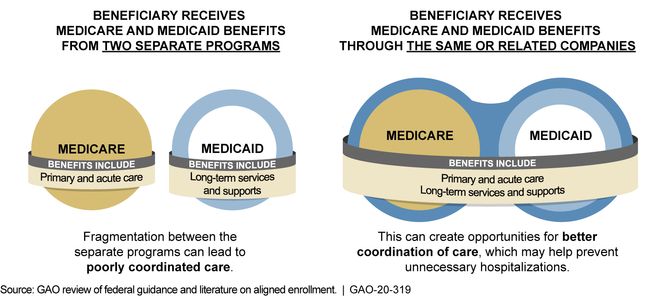
Dual-eligible beneficiaries are Medicare beneficiaries who are also enrolled in the Medicaid program in their state. In certain states, they may receive both types of benefits through private managed care plans. As of January 2019, about 386,000 such individuals were enrolled in both a private Medicare plan known as a dual-eligible special needs plan (D-SNP) and a Medicaid managed care organization (MCO) that were offered by the same or related companies. This arrangement, known as aligned enrollment, may create opportunities for better coordination between Medicare's acute care services and Medicaid's long-term services and supports, such as nursing facility care or personal care services.
Example of Aligned Enrollment through Managed Care for a Dual-Eligible Beneficiary
Medicaid officials in seven selected states described challenges with aligned enrollment. One challenge cited by officials in six of the states was using D-SNP and Medicare data to implement and evaluate aligned enrollment. For example, officials in one state said they cannot separate D-SNP quality data for just their state, because some D-SNPs report data spanning multiple states to the Centers for Medicare & Medicaid Services (CMS). As of December 2019, CMS officials said they are determining the best way for D-SNPs to report these quality data.
CMS has assisted states with aligned enrollment, but lacks quality information on the experiences of dual-eligible beneficiaries who have aligned enrollment through a process known as default enrollment. With default enrollment, states allow automatic assignment of beneficiaries who are enrolled in a Medicaid MCO and are about to become eligible for Medicare to the D-SNP aligned with that MCO. However, CMS's monthly reports on default enrollment do not include information on beneficiaries who choose to disenroll in the first 90 days after being default enrolled, a time frame specified in regulation. According to one beneficiary group, some beneficiaries may disenroll, because they did not realize they were default enrolled and their provider is not in the D-SNP's network. Quality information on the experiences of dual-eligible beneficiaries after default enrollment would allow CMS to better identify the extent to which beneficiaries face challenges and to determine how, if at all, to address the challenges.
Why GAO Did This Study
Congress authorized the establishment of D-SNPs in 2003 to address the unique needs of dual-eligible beneficiaries. For example, D-SNPs are required to provide certain specialized services targeted at the needs of dual-eligible beneficiaries, such as health risk assessments. D-SNPs must have approval of state Medicaid agencies to operate, and states can require D-SNPs to coordinate with Medicaid.
Congress included a provision in statute for GAO to review D-SNPs’ integration with state Medicaid programs. This report, among other objectives, (1) describes what is known about selected states’ experiences with aligned enrollment in D-SNPs, and (2) examines CMS’s oversight of aligned enrollment.
GAO reviewed relevant federal guidance and internal control standards. GAO also interviewed Medicaid officials in seven selected states and reviewed available documentation. The states (Arizona, Florida, Kansas, New Jersey, Pennsylvania, Tennessee, and Virginia) were selected, in part, for variation in experiences with aligned enrollment. GAO also interviewed officials from CMS, beneficiary groups, and companies that offered D-SNPs and Medicaid MCOs.
Recommendations
GAO recommends that CMS take steps to obtain quality information on the experiences of dual-eligible beneficiaries who have been default enrolled into D-SNPs. The Department of Health and Human Services concurred with the recommendation.
Recommendations for Executive Action
| Agency Affected | Recommendation | Status |
|---|---|---|
| Centers for Medicare & Medicaid Services | The Administrator of CMS should take steps to obtain quality information on the experiences of dual-eligible beneficiaries who have been default enrolled into D-SNPs, such as by obtaining information about the extent to which and reasons that beneficiaries disenroll from a D-SNP after being default enrolled. (Recommendation 1) |
In September 2021, CMS provided us with two new reports that give information on disenrollments of beneficiaries who have been default enrolled into D-SNPs. The reports include plan-level information on the number and percent of disenrollments from D-SNPs that are approved to conduct default enrollment. According to CMS officials, they plan to run these reports on a quarterly basis, and then review the data for trends at the plan level.
|
