Examining Ocean Acidification on World Oceans Day
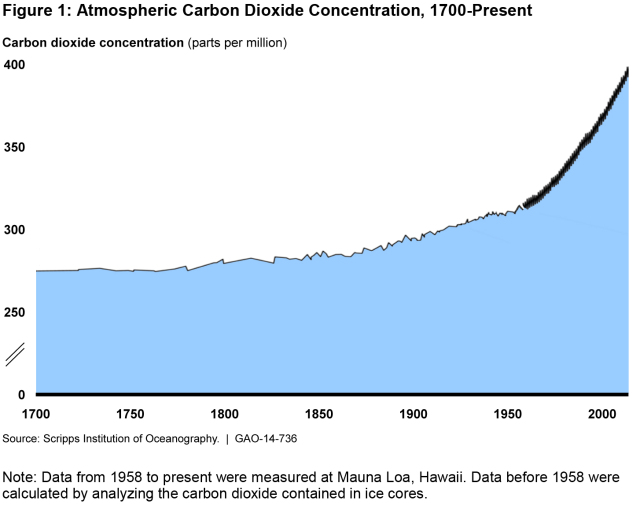 (Excerpted from GAO-14-736)
(Excerpted from GAO-14-736)
In addition to increasing acidity, the higher levels of carbon dioxide in the oceans cause chemical reactions that reduce what is known as the “saturation state” of certain calcium carbonate minerals. Lower “saturation states” mean some marine life will need to use more energy to build shells or skeletons.
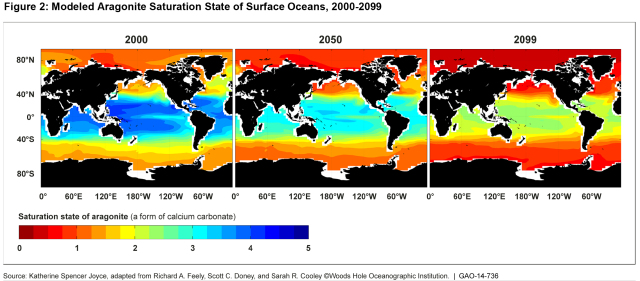 (Excerpted from GAO-14-736)
(Excerpted from GAO-14-736)
- Some marine species, such as oysters, may struggle to form or keep their shells, or have to alter their behavior, potentially affecting their survival.
- Marine ecosystems could change, altering, for example, predator and prey relationships.
- Coastal fishing and tourism industries could see changes in their economy.
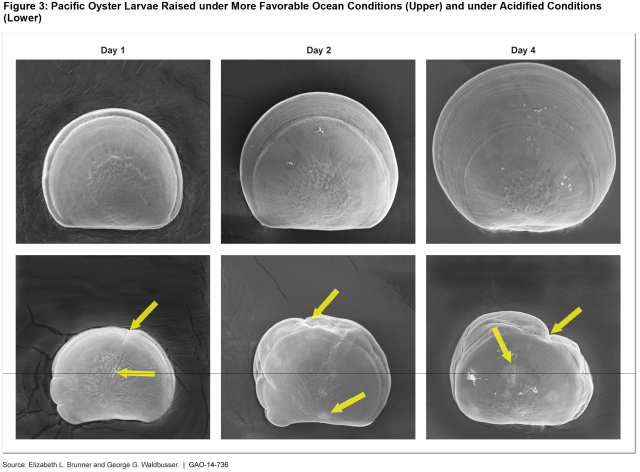 (Excerpted from GAO-14-736)
(Excerpted from GAO-14-736)
However, the scientific understanding of these effects is still developing, and there’s uncertainty about their scope and severity. For example, can species adapt to changes in ocean chemistry? One study we reviewed noted that it might be possible for some coral species to evolve a mechanism that would enable them to calcify at normal rates even in waters with lower carbonate ion concentrations, although this type of adaptation has not been documented in coral.
Federal efforts The Federal Ocean Acidification and Monitoring Act directed the National Oceanic and Atmospheric Administration and other federal agencies to take a number of steps to address ocean acidification. For example, NOAA and other agencies have- established an interagency working group composed of representatives from 11 federal agencies, including the EPA and USDA;
- developed a research and monitoring plan that includes using NASA satellites collecting data on ocean ecology and sharing that information with researchers; and
- supported relevant science, including multiple National Science Foundation-funded studies on changes to ocean chemistry.
- Questions on the content of this post? Contact Steve Morris at morriss@gao.gov.
- Comments on GAO’s WatchBlog? Contact blog@gao.gov.

GAO's mission is to provide Congress with fact-based, nonpartisan information that can help improve federal government performance and ensure accountability for the benefit of the American people. GAO launched its WatchBlog in January, 2014, as part of its continuing effort to reach its audiences—Congress and the American people—where they are currently looking for information.
The blog format allows GAO to provide a little more context about its work than it can offer on its other social media platforms. Posts will tie GAO work to current events and the news; show how GAO’s work is affecting agencies or legislation; highlight reports, testimonies, and issue areas where GAO does work; and provide information about GAO itself, among other things.
Please send any feedback on GAO's WatchBlog to blog@gao.gov.
