Public Health Preparedness: HHS Should Address Strategic National Stockpile Requirements and Inventory Risks
Fast Facts
The U.S. Strategic National Stockpile is a multibillion dollar inventory of medical countermeasures—medication, supplies, and more—that can be used in emergencies. COVID-19 underscored its importance.
The Department of Health and Human Services' inventory planning reports didn't meet most legal requirements enacted in 2019 or communicate risks associated with not meeting recommended inventory levels. This is partly because HHS hasn't updated its processes for completing the reports and a key advisory body was inactive.
Our recommendations address this.
HHS's leadership and coordination of public health emergencies is on our High Risk list.
Highlights
What GAO Found
The Strategic National Stockpile (SNS) is a multibillion dollar inventory of drugs, vaccines, supplies, and other medical countermeasures that can be used in emergencies. GAO reported in August 2022 that to guide inventory purchases from 2015 through 2019, the Department of Health and Human Services (HHS) used a multi-step process involving interagency experts, resulting in annual SNS reviews with inventory recommendations. This process was suspended when the expert group underwent a reorganization, and annual reviews were not completed to inform inventory decisions for fiscal years 2020 through 2022, resulting in purchases based on past reviews and HHS discretion. HHS has since completed reviews to inform inventory decisions for fiscal years 2023 and 2024. However, these reviews did not meet most statutory requirements—such as by including the amount of additional medical countermeasures procured—because HHS did not update its procedures to account for changes enacted in 2019. Until HHS updates its procedures, the agency risks not meeting the statutory requirements designed to give Congress additional information about the SNS inventory.
From fiscal years 2015 through 2021, HHS obligated nearly $5 billion in non COVID-19 appropriations to purchase medical countermeasures, mostly for anthrax and smallpox.
Obligations from Non COVID-19 Appropriations for Strategic National Stockpile from Fiscal Years 2015 through 2021

GAO's analysis of SNS reviews shows the SNS contained most medical countermeasure types recommended, but often not in the recommended quantities. HHS officials noted that gaps in quantities are due to budget constraints and acknowledge these gaps present risks. However, the reviews lack key information needed for managing these risks and communicating them to stakeholders, including to Congress. Risk management principles include guidance related to the management and communication of risk. Without an approach for regularly managing risks, HHS and Congress lack assurance the department is most effectively preparing for public health emergencies.
HHS obligated $6.1 billion in COVID-19 relief funds for supplies for the SNS that significantly increased the amount of certain medical countermeasures and expanded the types of countermeasures in the SNS inventory. For example, most of the funding went toward ventilators and personal protective equipment, which resulted in substantial increases in the amount of these medical countermeasures in the SNS relative to what was held prior to the COVID-19 pandemic.
Obligations Using COVID-19 Relief Funds for COVID-19 Supplies Delivered to the Strategic National Stockpile, Fiscal Years 2020 and 2021
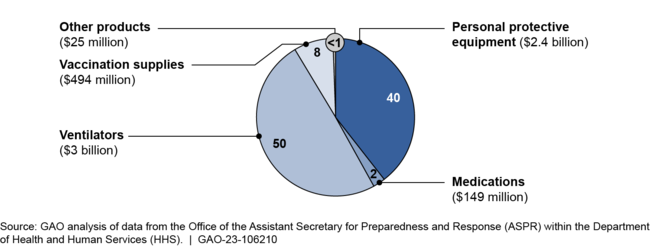
Additionally, the SNS inventory now contains additional finished pharmaceutical products, such as sedatives for use with ventilators. In response to recommendations from HHS, ASPR also took steps to add testing supplies to the SNS inventory in late 2020, including nasal swabs and transfer media. Prior to the COVID-19 pandemic, the SNS did not hold these medical countermeasures.
The COVID-19 response has also been a catalyst for HHS to re-examine SNS operations, including the role, responsibilities, expertise, and inventory needed moving forward. As such, HHS is working to develop a strategic plan to guide future SNS efforts.
Why GAO Did This Study
Recent emergencies have highlighted the importance of preparedness. One key component of the nation's medical response infrastructure is the SNS. The SNS inventory may be deployed to state, local, territorial, tribal, and international governments when needed. GAO placed HHS's leadership and coordination of public health emergencies on its High Risk List in January 2022 (GAO-22-105291) in part due to deficiencies in HHS's management of countermeasures.
The Pandemic and All-Hazards Preparedness and Advancing Innovation Act of 2019 included a provision for GAO to review the SNS. This report examines: (1) the process used to make inventory decisions; (2) non COVID-19 obligations for countermeasures and their alignment with recommendations; and (3) obligations for countermeasures using COVID-19 relief funds, and inventory and operations changes in response to the COVID-19 pandemic.
To conduct this work, GAO reviewed standard operating procedures, statutory requirements, inventory and obligations data, and other documentation; compared HHS actions to risk management practices; and interviewed HHS officials.
This report is a public version of a sensitive report issued in August 2022.
Recommendations
GAO is making three recommendations, including that HHS update procedures for SNS reviews and manage risks associated with inventory gaps. HHS concurred with GAO's recommendations.
Recommendations for Executive Action
| Agency Affected | Recommendation | Status |
|---|---|---|
| Office of the Assistant Secretary for Preparedness and Response | The Assistant Secretary for Preparedness and Response should update procedures for how SNS reviews will be conducted in accordance with statutory requirements, including a description of the roles and responsibilities of its interagency partners in the development of the SNS reviews. (Recommendation 1.) |
Open
In February 2023, the Department of Health and Human Services noted that ASPR had completed an initial draft of the SNS review that will inform inventory decisions for fiscal years 2024 and 2025, now known as the Medical Countermeasure Preparedness Review. HHS indicated that the review, once complete, would address relevant statutory requirements, consistent with our recommendation. Additionally, HHS noted that ASPR plans to update its procedures for conducting future reviews by summer 2023. They added that this update should include, among other things, a description of the roles and responsibilities for interagency partners in the SNS review process. We will continue to monitor this recommendation and update it as procedures are finalized.
|
| Office of the Assistant Secretary for Preparedness and Response | The Assistant Secretary for Preparedness and Response should develop and document an approach—whether through the standard operating procedures for the SNS reviews or some other mechanism—for ensuring that MCMs under consideration for SNS procurement receive the same consideration regardless of whether they received development funding from BARDA, in accordance with statutory requirements. (Recommendation 2.) |
Open
In Feb 2023, the Department of Health and Human Services noted that ASPR was in the process of updating its procedures for conducting SNS reviews consistent with past practice, with a planned completion by summer 2023. According to HHS officials, the update will involve an examination of medical countermeasures against several criteria, which will be applied regardless of whether the countermeasures received development funding from BARDA. We will update this recommendation after the new procedures are finalized. We maintain that the updated procedures should provide sufficient detail on how BARDA-supported products are considered for the SNS inventory to ensure those products are provided equal consideration, consistent with statutory requirements.
|
| Office of the Assistant Secretary for Preparedness and Response | The Assistant Secretary for Preparedness and Response should develop and document an approach for regularly managing the risks associated with the gaps between SNS MCM inventory levels and recommended quantities. Such an approach, which could occur as part of the SNS reviews, should clearly prioritize risks, track progress made in addressing the risks, and estimate resources needed to address risks. This approach should involve communicating this information to key decision makers, including Congress. (Recommendation 3.) |
Open – Partially Addressed
In February 2023, the Department of Health and Human Services noted that ASPR is developing and documenting a standard approach for managing risks associated with gaps in the SNS inventory. They added that ASPR has been in discussions with the Department of Homeland Security about best practices on risk prioritization for analyzing gaps against funding constraints. HHS noted that this risk prioritization framework will be incorporated into ASPR's procedures for conducting future SNS reviews, which the agency is in the process of updating and, at the time, expected to finalize by summer 2023. HHS added that the procedures will also outline an approach for conveying risk in an unclassified way, as much of the information related to risk prioritization is and must remain classified. In February 2024, the Department of Health and Human Services noted that leadership from the Public Health Emergency Medical Countermeasures Enterprise (PHEMCE)-an interagency effort responsible for defining and prioritizing requirements for public health emergency medical countermeasures-was routinely meeting to assess threats, evaluate and prioritize risks in conjunction with the Department of Homeland Security, and assess gaps in the SNS inventory. Additionally, HHS provided copies of the 2022-2026 PHEMCE Multi-Year Budget and the 2022 SNS annual review (now called the Medical Countermeasure Preparedness Review) which were delivered to Congress in March and May 2023, respectively. While these efforts demonstrate progress in managing risk-especially an active PHEMCE and regular communications with Congress on inventory and resources-we would like to see documentation that outlines how the PHEMCE risk management activities noted above are conducted. Until we see such documentation, it is unclear how risks, gaps, priorities, and progress are determined from year to year. HHS officials said that they are continuing to draft procedures but did not have a timeframe for completion. We will update this recommendation after we discuss HHS's approach with them and are able to review additional documentation.
|
